The density of aluminum is 2.70 grams per cubic centimeter (g/cm³). This value is equivalent to 2700 kilograms per cubic meter ( kg/m ³) and 0.0975 pounds per cubic inch (lb/in³). This low density, roughly one-third the density of steel, establishes aluminum as a primary lightweight metal in engineering and manufacturing. While this figure is standard for pure aluminum, the density varies slightly across different aluminum alloys, such as 6061 or 7075, depending on their specific composition. This guide provides a technical overview of aluminum's density, its comparison with other materials, and its practical implications in industrial applications.
What is Aluminum Density?
The Scientific Definition of Density
Density is a fundamental physical property of a material. It quantifies the mass of that substance contained within a specific unit of volume. As an intrinsic property, the density of a pure, uniform substance is constant regardless of the sample's size or shape. The relationship is expressed by the formula: Density = Mass / Volume.
Standard Density Value for Pure Aluminum
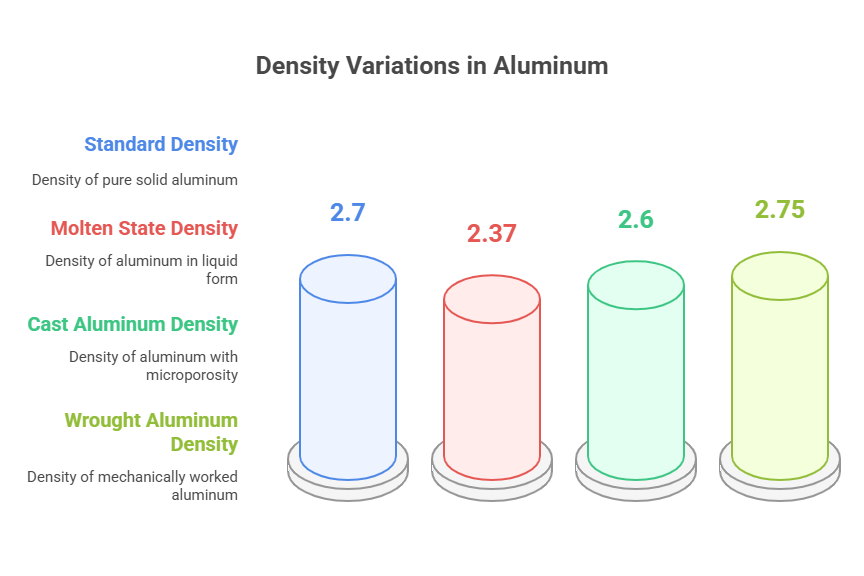
The accepted industry standard for the density of pure aluminum is 2.70 g/cm³ (grams per cubic centimeter). In the International System of Units (SI), this value is expressed as 2700 kg/m ³ (kilograms per cubic meter). These figures are standardized for aluminum at room temperature, typically 20°C (68°F). It is important to recognize that this value pertains to commercially pure aluminum, and will vary slightly with the introduction of alloying elements.
What is Specific Gravity?
Specific gravity is a ratio that compares the density of a substance to the density of a reference substance, which for solids is typically water at its densest (4°C). Because it is a ratio of two like quantities, specific gravity is a dimensionless value. For aluminum, the specific gravity is approximately 2.70. This figure is often used in engineering and material science tables to provide a quick reference for a material's weight relative to water.
How Light is Aluminum, Really?
To contextualize aluminum's classification as a lightweight material, a direct comparison of its density against other common engineering materials is necessary. Its advantage becomes evident when measured against both metallic and non-metallic alternatives.
Density Comparison with Common Metals
Aluminum's density is significantly lower than that of most other common metals, including steel, copper, and nickel. This difference is a primary driver for its selection in applications where minimizing weight is a priority. Magnesium is one of the few structural metals that is less dense than aluminum.
Table 1: Density Comparison of Common Metals
Material | Nominal Density (g/cm³) | Density Relative to Aluminum |
|---|---|---|
Magnesium | 1.74 | 0.64x |
Aluminum | 2.70 | 1.00x |
Titanium | 4.51 | 1.67x |
Zinc | 7.14 | 2.64x |
Carbon Steel | 7.85 | 2.91x |
Nickel | 8.90 | 3.30x |
Copper | 8.96 | 3.32x |
Density Comparison with Non-Metallic Materials
When compared to non-metallic materials, aluminum is denser than most plastics and some composite materials. However, its superior strength and stiffness give it a far higher strength-to-weight ratio than plastics, positioning it as a premier lightweight structural material.
Table 2: Density Comparison with Non-Metallic Materials
Material | Nominal Density (g/cm³) |
|---|---|
ABS Plastic | ~1.05 |
Nylon (Polyamide) | ~1.14 |
Carbon Fiber Composite (CFRP) | ~1.60 |
Glass (Soda-Lime) | ~2.50 |
Aluminum | 2.70 |
Do All Aluminum Alloys Have the Same Density?
No, the density of an aluminum alloy is not constant. The addition of alloying elements alters the material's final density from the baseline of pure aluminum.
How Alloying Elements Impact Density
The density of an aluminum alloy is a function of its chemical composition. When elements denser than aluminum, such as copper (8.96 g/cm³) or zinc (7.14 g/cm³), are added, the resulting alloy's density increases. Conversely, when lighter elements like magnesium (1.74 g/cm³) or lithium (0.53 g/cm³) are used as the primary alloying agent, the density of the alloy decreases.
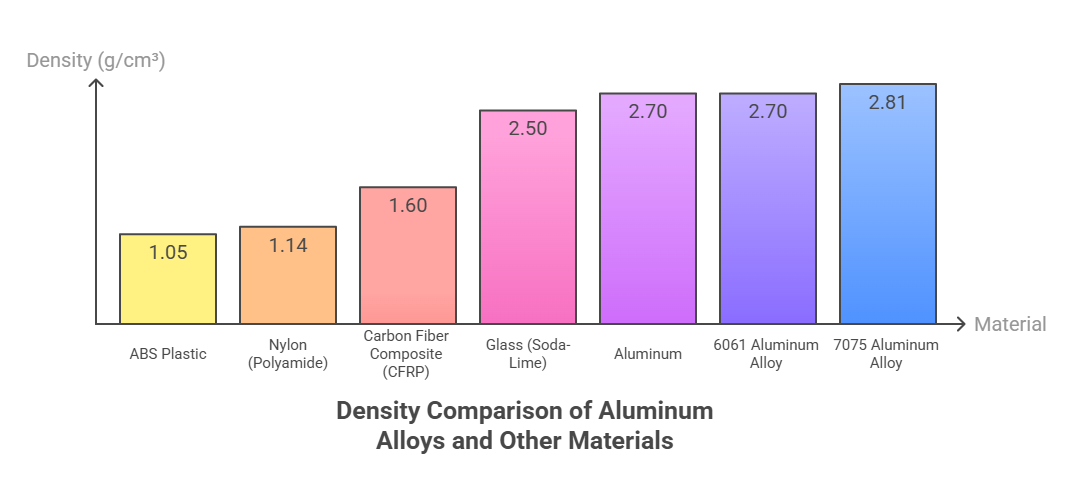
Density Variations Across Major Alloy Series
The industry-standard aluminum alloy designation system groups alloys into series based on their principal alloying elements. This results in predictable density ranges for each series.
Table 3: Typical Densities of Aluminum Alloy Series
Material | Nominal Density (g/cm³) | Typical Density Range (g/cm³) |
|---|---|---|
1xxx | None (99%+ Pure Al) | ~2.70 |
2xxx | Copper (Cu) | 2.75 - 2.84 |
3xxx | Manganese (Mn) | ~2.73 |
5xxx | Magnesium (Mg) | 2.65 - 2.68 |
6xxx | Magnesium (Mg) & Silicon (Si) | ~2.70 |
7xxx | Zinc (Zn) | 2.80 - 2.85 |
In-Depth Look: 6061 vs 7075 Density
A frequent comparison is made between 6061 and 7075, two of the most common heat-treatable alloys. 6061 aluminum alloy has a density of 2.70 g/cm³, identical to pure aluminum. In contrast, 7075 aluminum alloy has a higher density of 2.81 g/cm³. This increase is due to the high percentage of zinc in the 7075 composition. The trade-off for this marginal increase in weight is a substantial gain in strength, making 7075 a preferred material for high-stress aerospace applications.
What Factors Change Aluminum's Density?
While a standard density value exists for pure aluminum, this figure can be influenced by several external and internal factors. Temperature, physical state, and the manufacturing process can all induce variations in the material's measured density.
The Effect of Temperature
Like most materials, aluminum expands when heated and contracts when cooled. This phenomenon, known as thermal expansion, means that as the temperature of an aluminum component increases, its volume increases. Since density is mass divided by volume, an increase in volume results in a decrease in density. The linear coefficient of thermal expansion for aluminum is approximately 23 x 10⁻⁶ per degree Celsius (m/m/°C).
The Influence of Physical State
A material's physical state significantly affects its density. When aluminum is heated to its melting point (approximately 660°C or 1220°F), it transitions from a solid to a liquid. In its molten state, the density of aluminum decreases to approximately 2.37 g/cm³. This reduction occurs because the ordered crystalline structure of the solid state breaks down, and the atoms move more freely, occupying a larger volume.
Manufacturing Process: Cast vs. Wrought
The method used to form an aluminum part can influence its final bulk density. Cast aluminum alloys are produced by pouring molten metal into a mold. This process can sometimes trap microscopic gas bubbles or create shrinkage voids, a condition known as microporosity. This porosity means the part contains empty space, lowering its overall density compared to a perfectly solid piece of the same alloy. In contrast, wrought aluminum products are mechanically worked (e.g., rolled, extruded), which helps to close these internal voids, resulting in a more uniform and generally higher density that is closer to the material's theoretical value.
Why is Aluminum's Low Density Important?
Aluminum's low density is not merely a data point; it is the foundation of its utility across countless industries. This single property directly translates into significant performance and economic advantages.
The Strength-to-Weight Ratio Explained
The strength-to-weight ratio, or specific strength, is a measure of a material's strength divided by its density. Aluminum and its alloys exhibit a high strength-to-weight ratio. This means they can provide substantial structural integrity for a fraction of the weight of denser materials like steel. This characteristic is indispensable in industries like aerospace, where minimizing mass is paramount, and in high-performance automotive manufacturing, where it contributes to superior acceleration and handling.
Impact on Transportation and Fuel Efficiency
In the transportation sector, mass reduction is directly linked to fuel economy. Using aluminum components in vehicles—from chassis and body panels to engine blocks—lowers the overall vehicle weight. A lighter vehicle requires less energy to accelerate and maintain speed, which in turn reduces fuel consumption and lowers emissions. This principle applies to everything from commercial airliners to passenger cars.
Cost Implications in Logistics
The low density of aluminum yields tangible economic benefits in logistics. Lighter materials are less expensive to transport. Shipping costs are often calculated based on weight, so a shipment of aluminum parts will cost less than an equivalent shipment of steel parts. Furthermore, lighter components are easier and often safer for workers to handle and install, which can reduce labor costs and project timelines.
Relationship with Other Properties
Density also has an interesting relationship with some of aluminum's other key properties. For instance, consider electrical conductivity. While copper is a better electrical conductor by volume, aluminum is superior by weight. An aluminum cable can carry the same amount of electrical current as a copper cable that weighs twice as much. This is why aluminum is the dominant material for overhead power transmission lines.
How to Calculate the Weight of Aluminum?
Calculating the weight of an aluminum component is a straightforward process required for design, shipping, and cost estimation.
The Fundamental Calculation Formula
The weight of any object is found by multiplying its volume by its material density.
Weight = Volume × Density
Before performing the calculation, it is essential to ensure that the units for volume and density are consistent. For example, if density is in g/cm³, the volume must be calculated in cm³.
Step-by-Step Calculation Guides
Below are examples for common aluminum shapes. The density of 6061 aluminum (2.70 g/cm³) is used for these calculations.
Example 1: Calculating the Weight of an Aluminum Plate
- Dimensions: Length = 100 cm, Width = 50 cm, Thickness = 0.5 cm
Calculate Volume:
Volume = 100 cm × 50 cm × 0.5 cm = 2500 cm³
Calculate Weight:
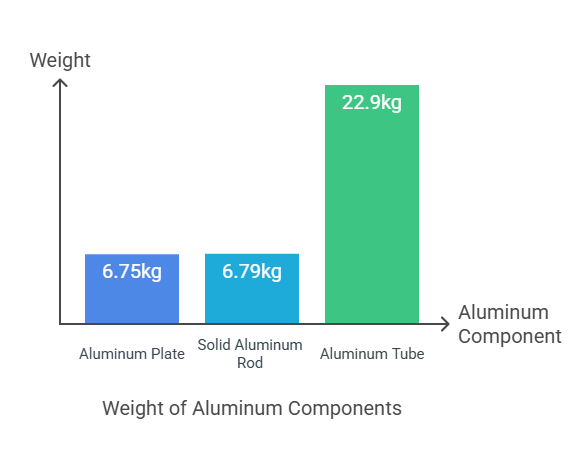
Weight = 2500 cm³ × 2.70 g/cm³ = 6750 g, or 6.75 kg
Example 2: Calculating the Weight of a Solid Aluminum Rod
- Dimensions: Length = 200 cm, Diameter = 4 cm (Radius = 2 cm)
Calculate Volume:
Volume = π × (radius)² × length = 3.14159 × (2 cm)² × 200 cm ≈ 2513.3 cm³
Calculate Weight:
Weight = 2513.3 cm³ × 2.70 g/cm³ = 6786 g, or 6.79 kg
Example 3: Calculating the Weight of an Aluminum Tube
- Dimensions: Length = 300 cm, Outer Diameter = 10 cm (R = 5 cm), Inner Diameter = 8 cm (r = 4 cm)
Calculate Cross-Sectional Area:
Area = π × (R² - r²) = 3.14159 × ((5 cm)² - (4 cm)²) = 3.14159 × (25 - 16) cm² = 28.27 cm²
Calculate Volume:
Volume = Area × length = 28.27 cm² × 300 cm = 8481 cm³
Calculate Weight:
Weight = 8481 cm³ × 2.70 g/cm³ = 22899 g, or 22.9 kg
A Note on Using Online Calculators
For quick estimations, numerous online metal weight calculators are available. While useful, users should exercise caution. Always double-check that you are inputting the dimensions in the correct units (e.g., inches vs. millimeters) and that the calculator is using the correct density value for the specific aluminum alloy in question.
How is Density Actually Measured?
The density values presented in technical literature are determined through precise measurement methods. These techniques are used both in laboratory settings for material characterization and on the production floor for quality assurance.
Standard Laboratory Methods
The most common and classic method for measuring the density of a solid object is the Archimedes' principle, also known as the water displacement method. This process involves three steps:
- The dry weight of the aluminum sample is measured using a precision scale.
- The sample is fully submerged in a fluid of known density (typically deionized water), and its "submerged weight" is measured. The difference between the dry weight and the submerged weight equals the weight of the displaced fluid.
- The volume of the sample is calculated from the weight and known density of the displaced fluid. Finally, the sample's density is determined by dividing its original dry weight by its calculated volume.
Quality Control in Manufacturing
In a manufacturing environment, density measurement serves as a critical quality control tool. For aluminum foundries, a quick density test on a sample from a new melt can verify its chemical composition. Since each alloying element has a distinct effect on density, a measured density that deviates from the expected value for a specific alloy can indicate an incorrect mixture or the presence of impurities. Furthermore, for cast products, comparing the measured bulk density of a part to the theoretical density of the alloy can reveal the level of internal porosity, providing insight into the quality and integrity of the casting.
Final Questions and Key Takeaways
This section addresses common questions related to aluminum's properties and summarizes the most important concepts covered in this guide.
Is Aluminum Considered a Heavy Metal?
There is no single, universally accepted definition that draws a clear line for what constitutes a heavy metal. From a chemical perspective, aluminum is sometimes classified as such based on its atomic weight. However, in the context of engineering, materials science, and industrial applications, aluminum is unequivocally considered a lightweight metal. The term "heavy metal" in this field is reserved for materials with significantly higher densities, such as steel, copper, or lead.
What is the Strongest Grade of Aluminum?
The strongest commercially available grades of aluminum are found in the 7000 series. Specifically, alloys like 7075 are renowned for their exceptional strength, which can rival that of some steels. It is worth noting the relationship between strength and density in this case. The high strength of the 7000 series is achieved by alloying with zinc, which, as previously discussed, increases the density of the alloy compared to pure aluminum. Therefore, the highest strength in aluminum often comes with a slight weight penalty.
Summary
- Standard Value: The benchmark density for pure aluminum is 2.70 g/cm³ (2700 kg/m ³).
- Key Advantage: Aluminum's primary benefit is its low density, which is approximately one-third that of steel, leading to a high strength-to-weight ratio.
- Alloy Variation: The addition of alloying elements alters density. Copper and zinc increase it, while magnesium decreases it. 7075 alloy is denser than 6061 alloy.
- Practical Calculation: The weight of an aluminum part is calculated by the formula Weight = Volume × Density.
- Context is Crucial: Factors such as temperature and manufacturing processes (casting vs. wrought) can influence the final measured density of an aluminum component.


