
Architects, designers, and builders often face a critical choice in materials, one that balances appearance with long-term performance. This frequently leads to a fundamental question about a classic alloy: does brass rust?
The direct answer is no. Rust, by definition, is the reddish-brown iron oxide that forms when iron or steel corrodes. Brass, an alloy of copper and zinc, lacks the necessary iron to undergo this specific chemical process.
Instead of rusting, brass corrodes. This corrosion manifests not as destructive flaking, but as a surface transformation known as tarnish. Over time, this process can develop into a stable, often desirable, layer called a patina. This distinction is not merely semantic; it is a critical piece of knowledge for material selection in the building industry, influencing everything from maintenance schedules to long-term aesthetic goals.
Rust vs. Corrosion: A Fundamental Distinction
In professional construction and design, precision in language reflects precision in practice. The distinction between rust and corrosion is a prime example. Confusing the two can lead to incorrect material specification and flawed maintenance strategies.
Why Brass Is Not Susceptible to Rust
Brass is immune to rust for a simple reason: it contains no significant amount of iron.
Rusting is a specific form of corrosion exclusive to ferrous metals like iron and carbon steel. It is the formation of hydrated iron oxide, a reddish-brown, brittle substance that flakes away from the surface. This flaking continually exposes new, unprotected metal to the environment, creating a cycle of progressive and often rapid deterioration. Because brass is a non-ferrous alloy composed of copper and zinc, the chemical reaction that creates rust cannot physically occur.

Defining the Correct Terms: Corrosion, Tarnish, and Patina
To discuss the surface changes of brass with professional accuracy, three terms are essential:
- Corrosion: This is the broad, scientific term for the gradual degradation of a material due to chemical reactions with its environment. It is the parent category under which all other terms fall.
- Tarnish: This describes the initial, superficial layer of corrosion on non-ferrous metals. It is the first stage of oxidation on brass, often appearing as a dulling of its original sheen or a darkening of its surface tone.
- Patina: This is the stable, mature layer that develops on the surface of brass after prolonged exposure. It is the final result of the corrosion process, a tightly bonded film that marks the material's equilibrium with its surroundings.
Patina Formation on Brass Surfaces
The graceful aging of a brass fixture is not a sign of failure. It is a predictable and often highly valued surface transformation. This process moves through distinct stages, culminating in a finish that is both protective and visually rich.
How Environmental Exposure Creates a Patina
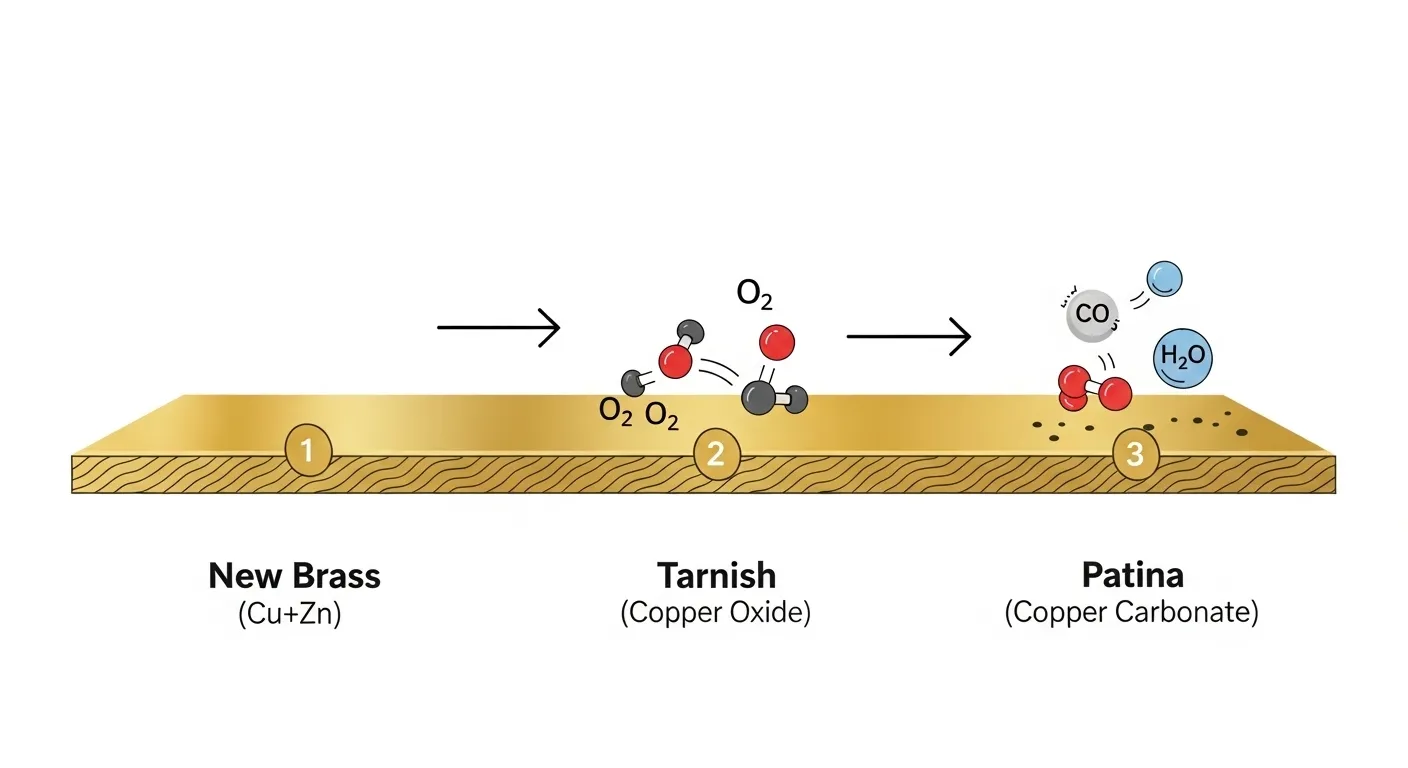
A newly installed brass component immediately begins a slow dialogue with its environment. The copper within the alloy reacts with atmospheric oxygen, creating an initial, dark layer of copper oxide. This is the tarnish stage.
As exposure continues, moisture, carbon dioxide, and other airborne elements like sulfur compounds contribute to a more complex series of chemical reactions. These reactions slowly convert the initial oxide layer into a more stable film of copper carbonate or copper sulfide. This mature, stable layer is the patina. Its final color, from deep bronze to the classic blue-green, is a direct reflection of the specific chemical makeup of its local environment.
The Nature of Patina: Protective Layer vs. Damage
The functional difference between the patina on brass and the rust on steel is absolute. One protects, the other destroys. For anyone specifying materials for longevity, this comparison is essential.
Characteristic | Rust (on Iron/Steel) | Patina (on Brass) |
|---|---|---|
Physical Structure | Porous, brittle, and flaky | Dense, adherent, and continuous |
Effect on Metal | A progressive, destructive cycle | A self-limiting, protective barrier |
Structural Impact | Weakens integrity, leads to failure | Preserves integrity, halts further corrosion |
Material Relationship | Parasitic: consumes the host metal | Symbiotic: shields the host metal |
Therefore, one process signifies material failure, while the other signifies the achievement of a stable, long-lasting state. The presence of a patina is evidence of the material's resilience, not its weakness. It is this self-preserving quality that allows brass to endure for generations in demanding architectural applications.
Factors Influencing the Rate of Brass Corrosion
The corrosion rate of brass is not a fixed property but a dynamic response to its environment. Its long-term performance and appearance are directly governed by exposure conditions.
The Impact of Water and High Humidity
Moisture acts as the primary electrolyte, accelerating the electrochemical reactions of corrosion. While atmospheric humidity drives a slow, natural aging process, prolonged water contact dramatically speeds up patina formation. In applications with constant water flow or immersion, a more aggressive form of corrosion known as dezincification can occur. This process leaches zinc from the alloy, leaving a weakened, porous copper structure, and is a critical failure consideration in plumbing systems.
Acceleration in Saltwater and Coastal Environments
The presence of chloride ions in marine air and saltwater creates a hyper-aggressive corrosive environment. Chlorides attack the zinc component in brass with extreme prejudice, causing rapid deterioration of standard alloys. For this reason, specifications for coastal or marine projects demand specialized materials like Naval Brass (C464), which includes tin to inhibit dezincification, or certain grades of bronze.

Considerations for Outdoor Architectural Applications
Specifying brass for exterior use is an exercise in predicting its environmental interaction.
- Climate dictates evolution: The same brass panel will remain bright for years in a dry climate but develop a deep green patina rapidly in a humid, polluted urban center.
- Alloy dictates character: The choice of alloy—from a yellow C260 Cartridge Brass to a reddish C230 Red Brass—determines the ultimate hue and texture of the mature patina.
- Intent dictates maintenance: A "living finish" that celebrates the patina requires minimal upkeep. A perpetually "new" finish requires a protective lacquer coating and a defined maintenance schedule from day one.
A Material Comparison: Brass vs. Other Metals
An effective material specification requires a comparative understanding of performance. The value of brass is best understood when juxtaposed with other common architectural metals.

Material | Primary Advantage | Corrosion Mechanism | Result of Corrosion |
|---|---|---|---|
Brass | Aesthetic & Workability | Forms a stable patina | Protective, often desired finish |
Bronze | Strength & Wear Resistance | Forms a stable patina | Protective, highly durable |
Copper | Conductivity & Malleability | Forms a prominent patina | Protective, archetypal green finish |
Carbon Steel | Cost & Strength | Forms destructive rust | Progressive material failure |
Stainless Steel | High Corrosion Resistance | Forms a passive oxide film | Highly resistant, but can pit in chloride environments |
Aluminum | Lightweight & Formability | Forms a passive oxide film | Highly resistant, but can corrode in extreme pH |
Maintenance and Care for Brass Fixtures and Components
The maintenance protocol for brass is dictated entirely by the desired aesthetic: a preserved polish or a developed patina.
Methods for Cleaning Tarnished Brass Surfaces
To restore a bright finish, the surface tarnish must be removed. This is a process of controlled abrasion and chemical action.
- Assess the Object: First, confirm it is solid brass (a magnet will not stick) and unlacquered. If a clear coat is peeling, it must be stripped professionally before polishing.
- Use a Dedicated Polish: Commercial brass polishes are the industry standard. They provide a consistent, controlled abrasive action that removes the copper oxide layer without damaging the underlying metal. Always follow the manufacturer’s directions precisely.
- Avoid Destructive Methods: Never use steel wool, scouring powders, or overly aggressive abrasives. These tools will permanently scratch the surface, creating a finish that looks dull even when clean.

Techniques for Preserving the Finish and Preventing Tarnish
Maintaining a polished look requires creating a barrier between the brass and the environment.
- Clear Lacquer Coating: For architectural elements, a professionally applied clear coat (often acrylic or urethane-based) is the most durable solution. This transparent film isolates the brass from atmospheric oxygen and moisture.
- Microcrystalline Wax: For interior objects or hardware subject to frequent handling, a thin layer of microcrystalline wax provides a renewable, invisible barrier against fingerprints and humidity. It is the standard for museum conservation.
- Routine Wiping: A simple, regular wipe with a dry, soft cloth can significantly slow tarnish formation by removing corrosive dust and moisture deposits.
FAQ Section
How long does it take for brass to develop a patina?
The timeline varies dramatically based on the environment. In a protected indoor setting, it can take years. In a coastal or industrial outdoor location, a noticeable patina can form within a few months to a year.
Is the green patina on brass harmful to touch?
No, the stable, solid patina on architectural brass is generally not harmful to touch in passing. It is primarily copper carbonate, which is not readily absorbed through the skin. However, it is not intended for food contact surfaces.
Can you stop brass from changing color completely?
Yes, by applying a protective barrier. A professionally applied clear lacquer or polyurethane coating will seal the brass from the atmosphere, effectively preventing oxidation and preserving its original polished finish.
Does polishing brass weaken it over time?
Excessive or aggressive polishing can cause microscopic wear over many decades, but standard, periodic cleaning with a proper brass polish will have a negligible structural impact on a solid brass component during its functional lifespan.
What is the difference between architectural bronze and brass?
While often used interchangeably in marketing, "architectural bronze" (like C385) is technically a type of brass, not a true bronze. It is a copper-zinc alloy containing lead to improve machinability. True bronzes are primarily copper-tin alloys.


